Hetrick, Dynamics of Nuclear Reactors, American Nuclear Society, 1993, ISBN: 3-2. 
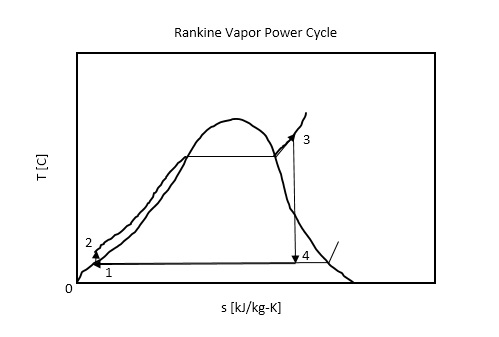
Neuhold, Introductory Nuclear Reactor Dynamics, American Nuclear Society, 1985, ISBN: 9-4. Bezella, Introductory Nuclear Reactor Statics, American Nuclear Society, Revised edition (1989), 1989, ISBN: 3-2. Department of Energy, Nuclear Physics and Reactor Theory. DOE Fundamentals Handbook, Volume 1 and 2. January 1993. Robert Reed Burn, Introduction to Nuclear Reactor Operation, 1988.Physics of Nuclear Kinetics. Addison-Wesley Pub. Nuclear and Particle Physics. Clarendon Press 1 edition, 1991, ISBN: 978-0198520467 Nuclear Reactor Engineering: Reactor Systems Engineering, Springer 4th edition, 1994, ISBN: 978-0412985317 Stacey, Nuclear Reactor Physics, John Wiley & Sons, 2001, ISBN: 0- 471-39127-1. Baratta, Introduction to Nuclear Engineering, 3d ed., Prentice-Hall, 2001, ISBN: 8-1. Lamarsh, Introduction to Nuclear Reactor Theory, 2nd ed., Addison-Wesley, Reading, MA (1983). The thermodynamic efficiency of this cycle can be calculated by the following formula: Where the temperature of the hot reservoir is 275.6☌ (548.7 K), the temperature of the cold reservoir is 41.5☌ (314.7K). In the ideal case (no friction, reversible processes, perfect design), this heat engine would have a Carnot efficiency of In this case, steam generators, steam turbines, condensers, and feedwater pumps constitute a heat engine subject to the efficiency limitations imposed by the second law of thermodynamics. On the other hand, most of the heat added is for the enthalpy of vaporization (i.e., for the phase change). 
Note that there is no heat regeneration in this cycle. The enthalpy difference between (2 → 3), which corresponds to the net heat added in the steam generator, is simply: For this entropy s 2 = 0.592 kJ/kgK and p 2 = 6.0 MPa we find h 2, subcooled in steam tables for compressed water (using interpolation between two states). State 2 is fixed by the pressure p 2 = 6.0 MPa and the fact that the specific entropy is constant for the isentropic compression (s 1 = s 2 = 0.592 kJ/kgK for 0.008 MPa). 174 = 1787 + 53.2 = 1840 kJ/kgĮnthalpy for state 1 can be picked directly from steam tables: The enthalpy for the state 3 can be picked directly from steam tables, whereas the enthalpy for the state 4 must be calculated using vapor quality: S l = entropy of saturated liquid water (J/kg K) = 0.592 kJ/kgK (for 0.008 MPa) S v = entropy of “dry” steam (J/kg K) = 8.227 kJ/kgK (for 0.008 MPa) S 4 = entropy of wet steam (J/kg K) = 5.89 kJ/kgK In the case of wet steam, the actual entropy can be calculated with the vapor quality, x, and the specific entropies of saturated liquid water and dry steam: The specific entropy of saturated liquid water (x=0) and dry steam (x=1) can be picked from steam tables. State 4 is fixed by the pressure p 4 = 0.008 MPa and the fact that the specific entropy is constant for the isentropic expansion (s 3 = s 4 = 5.89 kJ/kgK for 6 MPa). Since we do not know the exact vapor quality of the outlet steam, we have to determine this parameter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
